Wearable robotics designed to be used as a rehabilitation or augmentation medical devices is the second oldest field of exoskeleton development. The first working medical exoskeleton was created in 1972 by the Mihajlo Pupin Institute in Belgrade, Yugoslavia (modern day Serbia). Although several labs demonstrated early success with exoskeleton development for medical use, research in industrial robotics was far more practical and profitable. Decades later exoskeleton development was restarted initially to assist medical professionals, specifically for lifting and carrying patients.
Overtime, research focus shifted from medical professionals back to the patients themselves. One major breakthrough was the recognition that exoskeletons can perfectly recreate the same motion repeatedly thousands of times. This translates to patients being able to perform more exercise repetitions in the same amount of time with higher consistency. As a patient becomes stronger and more confident, the amount of assistance is dialed back. Ideally the required assistance reaches zero at the end of the rehabilitation program and the patient will never need to use the exoskeleton ever again.
This creates the first big split in medical wearable devices: rehabilitation vs. mobility aid. A rehabilitation device is used as a tool to augment a physical rehabilitation program and is no longer used after the completion of the program. A mobility aid wearable will be used permanently and the user is not expected to make a recovery. In this case the exoskeleton becomes an augmentative device.
Medical Exoskeletons for Rehabilitation vs Augmentation
The field of medical exoskeletons is by far the most varied and colorful of all of the subfields in the industry. In order to provide some structure, we have to divide what is currently being developed into broad categories. As mentioned above, medical exoskeletons and orthotics can be divided into two major groups: rehabilitative and augmentative. Rehabilitation medical exoskeletons assume that the user will improve after using the wearable device. After a supervised training regimen set, the user will no longer need to use the rehabilitation exoskeleton. An augmentative medical exoskeleton assumes that the user will not get better after undergoing rehabilitation and they will rely on the wearable device for the rest of their lives.
Here is a practical example of separating an exoskeleton for rehabilitation vs. one for augmentation / mobility aid: Ekso GT vs REX. The Ekso GT by Ekso Bionics has to be used by a person that still has some mobility and the user has to be able to shift weight from one leg to the other. The Ekso GT variable assist software allows for targeted assistance (one leg might need more assistance than the other) and a gradual decrease in the assistive force as the patient becomes stronger during rehabilitation. In contrast, the REX by REX Bionics takes full control of walking, including transferring weight from one leg to the other. The REX operator “rides” the suit and does not use their own muscles to walk. This results in none to minimum rehabilitation benefit*, however a person with complete paralysis can use it to stand up and walk. Thus while the Ekso GT and REX are both “medical exoskeletons,” they are aimed at patients with different needs and capabilities.
*Update Feb 2017: New research has demonstrated that exoskeletons which supply 100% of the needed power to move still have rehabilitation advantages.
Medical orthotics for rehabilitation vs. augmentation face different design challenges. A rehabilitation exoskeleton has be adjustable, execute the exact motion hundreds of times, log the information for each patient and the exo is used in 1 to 2 hour sessions per user. An augmentative exoskeleton needs to be fitted only for one user, needs to have longer battery life (if powered) and needs to be comfortable to wear for the entire day.
Comfort over long periods of time is an especially big challenge for augmentative / mobility assist exoskeletons. While wearables have made great strides in their size and usefulness, wearing a robot for many hours and trying to do everyday tasks remains a challenge. For example, having a powered glove to augment grip strength can be the difference between being able to use that hand and not, until it is time to clean the dishes or wash your hands in which case the powered glove can become a liability.
Upper Body vs Lower Body Medical Exoskeletons
Another way to differentiate medical exoskeletons is by the region they target, either the upper or lower body. Lower body medical exoskeletons aim to improve walking or aid individual joints (such as a knee only medical exoskeleton). Upper body medical orthotics focus on the arm (shoulder, elbow) or hand (fingers, wrist) strengthening or augmentation. Again, upper and lower body medical wearable robotics can still be divided into ones for rehabilitation vs. augmentation. In the past there were medical exoskeletons that targeted the upper and lower parts of the body simultaneously but few to none are currently left in development.
Stationary vs Mobile
A third way to categorize medical exoskeletons is by their size and mobility. Exos can be either stationary, tethered or mobile. Stationary exoskeletons are usually bolted to a wall or connected directly to the ground through a rigid frame. Tethered exoskeletons can be suspended on an overhead guide rail, supported by a metal frame on wheels or the exoskeleton can even be directly supported by a mobile robot. Stationary and tethered exoskeletons can have large motors and controllers and none of their weight will be distributed on the user. Mobile medical exoskeletons can be worn outside or in a person’s home. They require a battery pack and lighter motors and controllers that are fitted in a limited, compact space. This makes mobile exoskeletons much more complicated and expensive to design.
Control Types: pre-programmed to mind control and everything in between
A fourth way to characterize medical exoskeletons is on the control strategy that they use. The most basic strategy is a pre-programmed wearable robot that executes predetermined motions. A more complicated device would wait for a set of conditions to be met (example detection of shift of center of mass) before executing a motion. Even more complicated medical exoskeletons read electrical nerve signals from the spine, arms and legs and produce a proportional motor activation. Some medical exoskeletons are becoming better at reading the nerve signals directly from the brain to activate the medical wearable robot. The most complicated medical exoskeletons can provide FES (Functional Electrical Stimulation) while still controlling the exoskeleton.
Other Categories
There are many other categories that can be used to separate medical exoskeletons. For example, exos can be passive (no motors) vs. powered. Also, exoskeletons can be hindering such as a tremor suppression exoskeleton which provides motion opposite of the one caused during a tremor. Yet another category would be pediatric medical exos. Most exoskeleton just aren’t adaptable enough to fit children so a specialized pediatric version has to be designed and manufactured. In addition, many medical wearables are tied to screens and computer games to make completing repetitive tasks more entertaining and rewarding. Yet another way to categorize medical exos would be if they are rigid or made entirely out of soft materials. There is really no established correct way to categorize medical exos. However as the exoskeleton industry is growing and maturing some nomenclature convention will have to eventually be adopted.
For the purposes of this article the medical exoskeletons will be divided using only the first three of the categories described above: rehabilitative vs augmentative, upper body vs lower body and mobile vs stationary.
The following is a list of commercial medical exoskeletons, research projects that are close to becoming commercial products and research projects that have had a great influence on the industry. The devices are separated into the categories described earlier. Note that this list focuses on commercial or more famous medical exoskeletons. There are many research labs and companies with wonderful prototypes and products in various stages of development that have been omitted.
Stationary Lower Body Exoskeletons
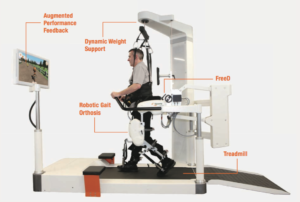
- Lokomat by Hocoma (optional parts for pediatric care)
- RoboGait by Bama Teknoloji
- InMotion Ankle by Interactive Motion Technologies (now purchased by Bionik Labs)
- Alex 3 by University of Delaware
- ANKLEBOT by MIT
Stationary Upper Body – Arm&Wrist Exoskeletons For Rehabilitation
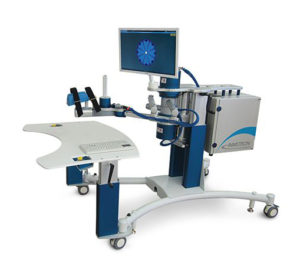
- InMotion Arm by Interactive Motion Technologies (now purchased by Bionik Labs)
- InMotion Wrist by Interactive Motion Technologies (now purchased by Bionik Labs)
- Armeo by Hocoma
- ALEx by KineteK Wearable Robotics
- Tack-Hold by KineteK Wearable Robotics
- Power Jacket REALIVE – Panasonic ActiveLink
- HARMONY – ReNeu Robotics Lab, UT Austin
Stationary Upper Body – Hand Exoskeletons For Rehabilitation

- Amadeo by Tyromotion
- InMotion Hand by Interactive Motion Technologies (now purchased by Bionik Labs)
- Hand of Hope by Rehab-Robotics
Mobile Upper Body – Exoskeletons For Rehabilitation & Augmentation

- MyoPro Motion G – Myomo
- CARAPACE by Lorenzo Masia
- Robotic Soft Extra Muscle (SEM) Glove by Bioservo
- Pneumatic Power Assist Glove by Daiya
- Inflatable Soft Exoskeleton by Otherlab Orthotics
- Inflatable Soft Robotic Glove – Wyss Institute
- Affordable Tremor Suppression Arm – MedEXO Robotics
Mobile Lower Body Rehabilitation Exoskeletons

- HANK by Gogoa
- ReWalk by ReWalk Robotics
- Hal Medical by CYBERDYNE
- Ekso GT by Ekso Bionics
- Indego by Parker Hannifin
- ExoAtlet by ExoAtlet
- ARKE by Bionik Labs
- Phoenix by SuitX (formally US Bionics)
- Atlas by Marsi Bionics
- MODO by Active Bionics
- Bionic Leg by AlterG
- AxoSuit by AxoSuit
- X1 Mina Exoskeleton by NASA-IHMC
- REX by REX Bionics

Mobile Lower Body Augmentation Exoskeletons
- REX P by REX Bionics
- Keeogo by B-Temia
- C-Brace by Ottobock
- Stride Management by Honda
- Body Weight Assist by Honda
- Exo-Legs PhaseX AB
- MAXX by ETH Zurich, Sensory-Motor Systems Lab
If you have read the sister article to this one, 22 Exoskeletons For Work and Industry Into 6 Categories you might have noticed that some of the exoskeletons are repeated. A handful of companies are marketing the same product for different applications and seeing what sticks. B-Temia for example is testing the waters for its knee exoskeleton, the Keeogo for the medical augmentative, military and consumer/civilian markets at the same time!



Your article helped me a lot thanks for your efforts.
I would like to add something up in the
”Stationary Lower Body Exoskeletons” section.
The products i found worth some time to look into them.
Motorika – Reo Ambulator
Reha Technology – GEO System
Meditouch – Balance Tutor
The previous companies have also stations for upper extremity so you add them up to some other sections also.
Kind regards
Thank you so much for contributing. I was vaguely aware of only one of the three companies you listed. As you say, they offer more than just gait rehabilitation solutions.
An interesting article, but I don’t agree with your comment about REX that “This results in none to minimum rehabilitation benefit,…”. See http://www.rexbionics.com/clinical-trials/ to find out more about the potential rehabilitation benefits of REX.
Thank you for Charles for your input. This article is starting to show its age. There are multiple studies that have demonstrated a benefit to getting people up on their feet and moving early.
Hi! I was wondering if you have references for your information?
Hi Curry,
There should be a link to every manufacturer’s website in each catalog entry. In addition, the majority of our articles also have references. The team wants to the Exoskeleton Report to be a web-portal in the world of real exoskeleton devices and for the audience to be able to quickly reach more in-depth information on every topic.