Description
Key attributes:
- Approved by the FDA for clinical and home use for individuals with spinal cord injury (SPI).
- Battery powered hip-knee exoskeleton.
- Proven to work in clinical, home and city environments.
- Regularly being improved and iterated (the 2016 ReWalk is now 6th revision)
- Controlled by subtle changes in the user’s center of gravity.
- Graphical user interphase for the physiotherapist’s control.
- Structured training program for the rehabilitation teams purchasing the device.
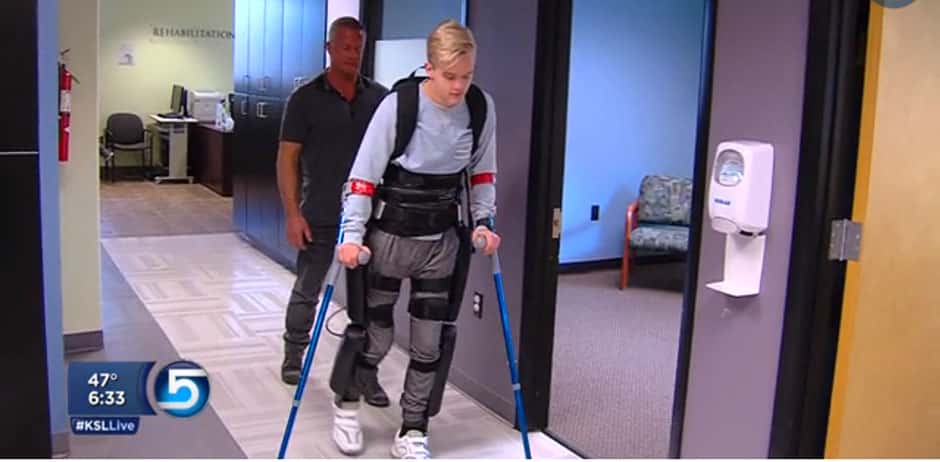
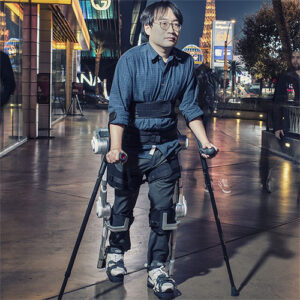
The ReWalk Rehabilitation System is optimized for use in a clinical setting. It is used as a tool to augment the capabilities of regular physiotherapy by providing a high number of consistent and reproducible steps. ReWalk Rehabilitation is also used as a stepping stone towards individuals preparing to acquire ReWalk for personal use.
ReWalk for personal use is quickly becoming a popular supplemental device to the wheelchair. There are ReWalk users that have been using their unit continuously twice a week (on average) for the last five years. Those users have become quite comfortable and proficient with their exoskeletons.
Lifeward, formerly ReWalk Robotics (and Argo Medical Technologies before), Offices in Massachusetts, USA; Berlin, Germany; Yokneam Ilit, Israel, website
Exoskeleton Report does not endorse one exoskeleton product over another. The exoskeleton catalog is purely for educational purposes. The catalog is meant to provide an easily accessible birds-eye view of the exoskeleton industry, and a quick method to sort exoskeletons by type and purpose. All prices are approximate and are meant to provide a general sense of the cost of the devices.















A12BMB –
Looking Forward for the Joint Venture with Realcan and the approval of the new Soft Exo suit.
Laura –
My family, physicians and Therapist were definitely misrepresented by this company! We were told that insurance would cover this equipment but it may take several months. But in reality.. only 1 insurance contact pays for the equipment, Cigna which was approved in February 2019. My son and our family were lead to believe in July 2018 that his it would be covered by BCBS. A representative of this company contacted us finally in January 2019 and told us that she was being flown in from Boston just to fit my son for this equipment. The representative communicated through her travels letting us know exactly where her flight landed and she was in our city. She fitted my son and did a trial run with the equipment! We were told he should have his own device within a few months! 7 months later we received a call starting it was denied by our insurance, so oh well he can’t get! I contacted both of my insurance companies and was told by both that a request was NEVER submitted by Rewalk!! This company lied to my son, my family, his physicians, his therapist and his community!! I feel like the company SCAMMED us!! They need to be exposed for giving false hope to paralyzed patients!
Bobby Marinov –
Dear Laura, I am heartbroken to hear what happened to you and your family. I encourage you to read what our team has written on both false-hope and Cigna. For example, to the best of my understanding, Cigna does not “pay for the equipment” they are supposed to have a policy that they will review each case individually.
Laura –
This company gives false hope to the most vulnerable patients
Laura Spivey –
Thank you very much for your concern. I’m trying to file an appeal with both of my insurance companies but I’m told that Rewalk never submitted a claim, an request or even called in a reference for the product! A person can not file an appeal if the company never submitted a request for the product? It doesn’t make sense! A representative flew from Boston to NC just to fit my son for the product. Why if the company knew it wasn’t covered by his insurance companies. I have emails showing that I obtained the order from his MD and letter of medical necessity as requested by the representative of the company. Why if they knew they were never going to file a claim or request prior approval?? It’s unfortunate that my son’s only hope for ever walking again was crushed by miscommunication of this company! He works everyday now.. saving every dollar he makes.. in hopes to one day buying this product! It breaks my heart and should break the hearts of any parent or any member of the Rewalk company!!
ReWalk –
ReWalk has not claimed any US insurer has a policy to cover the exoskeleton device. We do submit – when plans allow – for prior authorizations on a case by case basis. The press release related to the Cigna policy is pasted here, reflecting that:
https://rewalk.com/rewalk-announces-cigna-as-first-private-us-insurer-to-adopt-national-policy-change-for-coverage-of-personal-exoskeleton-devices/
When discussing ReWalk ownership with a candidate we give an overview of the process, in which we determine if the person is a candidate via a physical evaluation and also perform a benefits analysis. In some cases we can submit to the insurer, in others the insurer will not even accept an application for prior authorization due to ReWalk not being contracted with the insurer, or the insurer has a negative coverage policy. Naturally, we would not share a device candidate’s personal information or situation in this forum, but these are common causes of inability to submit for a device on a candidate’s behalf – which ReWalk does at no cost to them.
Unfortunately case by case coverage for any device is unpredictable and generates disappointment all too often. That being said over 170 ReWalk devices have been paid for by third parties, in addition to success in the US VA system and inclusion on the official list of medical aides in Germany. The disinformation on this topic is frustrating for all, so we strive to be upfront and avoid false hope. We never promise coverage, nor do we rule it out. We say we will submit when we can, whether the insurer accepts it is their decision.
Disinformation can also be harmful to others. We recently saw a post on a YouTube video which featured a ReWalker who received his device from his insurer after a work injury. A poster wrote that the video was untrue because without a HCPCS code an insurer cannot accept a submission. The poster in effect called the device recipient – a retired police officer who was injured in the line of duty – a liar despite viewing a video of the person walking in their own device.
This is just one example of the issues caused by the complexity and unpredictability of insurance coverage of newer devices, and the misinformation spread by uninformed parties.
We are hopeful for more systematic coverage going forward. As of now there is not a HCPCS code for any exoskeleton device so we cannot submit for Medicare coverage, but we did announce on our recent earnings call that we have submitted an application for one. We will also continue to engage payors with the goals of removing negative coverage policies and creating positive policies for coverage.
Any candidate can visit ReWalk.com/contact and we can discuss the physical criteria for using ReWalk and discuss their potential coverage pathway.
Andy –
As a quick update to the previous post – a HCPCS code was issued in July, 2020 which is the first to describe an exoskeleton. While this doesn’t guarantee coverage, it is a major step towards seeking policies for Medicare beneficiaries. Read more about this major development for the exoskeleton industry here – https://rewalk.com/centers-for-medicare-medicaid-services-issues-code-for-rewalk-personal-exoskeleton/
Kathryn –
I’ve seen this technology have a meaningful impact for many individuals. While walking is exciting in itself, the potential to provide health benefits is what gets individuals really committed!
Artin –
I am disabled and in a wheelchair, and I live in Canada. Mafia gangs spend millions of dollars on advertising, but they don’t value disabled people, especially lawyers, courts, and most importantly, the insurance mafia, and because I have no hope for the future, and my monthly income is so low for a family of 3 that I am ashamed. From my wife and son. And I hope to die soon. So that they at least feel free. Not that they try to take care of me all their life. I am 44 years old and I had an accident at the age of 37