Description
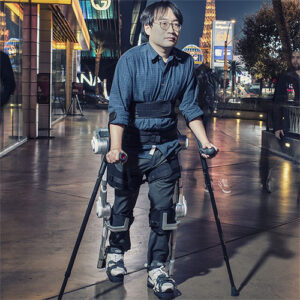
ABLE Exoskeleton is the rehabilitation tool for every clinic: it is meant to be the most lightweight and accessible hip-knee powered exoskeleton for neurorehabilitation with adjustable sizes. It actively assists individuals with mobility impairments to stand, walk, turn, and sit down.
The device has been validated in leading European hospitals like Heidelberg University Hospital (Germany), Institut Guttmann (Spain), and Sint Maartenskliniek (Netherlands), demonstrating its safety, feasibility, usability, and performance. Results showing improvements in clinical outcomes and benefits in rehabilitation are published in high-impact scientific journals (NATURE Scientific Reports, Journal of NeuroEngineering and Rehabilitation).
ABLE Exoskeleton offers unique characteristics in terms of:
● Physiological gait: helps re-teach the brain and muscles how to walk again.
● Quick donning: three times faster fitting, maximizing therapy time.
● Versatility: intuitive use, adapts to each patient’s specific needs across rehab.
● Cost-effectiveness: enables widespread clinical access to robotic rehabilitation.
This wearable device connects to a mobile app for personalized and data-driven therapy, helping therapists securely store session data, adjust exoskeleton settings, quantify gait performance in real time, and track each patient’s progress.
The ABLE Exoskeleton is intended to enable individuals with spinal cord injury at levels from C5 to L5 (with elbow extensor muscle activity of at least 4/5 in both arms) to perform ambulatory functions in rehabilitation institutions under the supervision of a trained therapist. It fits people 150-190 cm tall, weighing up to 100 kg.
Certifications:
In April, 2024, the company announced that it has received a CE Mark 2797 under the new Medical Device Regulation (MDR) for the ABLE Exoskeleton. This allows for the commercialization of the wearable device to hospitals and rehabilitation centers across the European Union.
“ABLE Exoskeleton’s technology is the fruit of hundreds of sessions with patients and therapists. Every single feature comes from attentively listening to what our users’ problems are and creatively solving them. From the mechanical design, to the electronic boards, the software, firmware or the control strategies, everything has been developed in-house by a remarkable group of engineering professionals”, said Alex Garcia, CTO of ABLE Human Motion. (source)
ABLE Exoskeleton – ABLE Human Motion, Calle Marie Curie, 8, 08042 Barcelona, Spain – website

Exoskeleton Report does not endorse one exoskeleton product over another. The exoskeleton catalog is purely for educational purposes. The catalog provides an easily accessible birds-eye view of the exoskeleton industry and a quick method to sort exoskeletons by type and purpose. All prices are approximate and are meant to provide a general sense of the cost of the devices.













Reviews
There are no reviews yet.