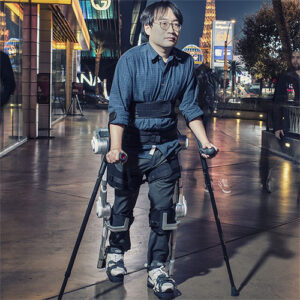
Description
The Ekso Indego® can be used for therapy as a gait training tool. The Indego Personal can also be employed as a supplementary mobility aid to a wheelchair. In addition to being able to see people eye-to-eye, research using this and other assistive medical exoskeletons has shown a strong correlation between standing up and a multitude of secondary benefits. These positive effects include but are not limited to improved bowel control, increased bone density, and reduction in pressure sores.
The Ekso Indego exoskeleton features a modular design and can be taken apart for easy storage and transportation. It is also the first exoskeleton to feature an iOS pad preloaded on an iPod Touch. The app is designed to provide effortless tracking of the user’s utilization of the wearable, export the data, and change the device settings. Both the Indego Therapy and Indego Personal have a CE Mark, allowing it to be sold commercially in Europe and the U.S. Also, the Indego has been approved by the FDA for clinical and personal use for patients with spinal cord injuries.
In the U.S., the Indego is marketed for individuals with complete and incomplete spinal cord injury T4 or below. For Europe, it is sold for individuals with lower limb weakness or paralysis (note: due to various regulations in North America, Europe, and Asia, it is widespread for exoskeleton devices to have different listed capabilities).
Indego design specifications:
- Hip-knee powered exoskeleton
- Li-ion batteries
- Total assembled device weight of 12kg (26lb)
- Modular
- Ability to export data using an iOS app
- Bluetooth connectivity
- Color LED and vibration user feedback (note: Recent independent research has shown haptic feedback to be instrumental in some gait rehabilitation cases)
- Built-in functional electrical stimulation (FES) interface (select models)
User requirements:
- Height: 155 to 191 cm (5’1” to 6’3”)
- Maximum weight: 113 kg (250 lb)
- Maximum hip width: 42.2 cm (16.6”)
- Femur length: 35 to 47 cm (14”-18.5”)
- Spasticity score: Modified Ashworth Score 3 or lower
- Enough upper body strength to balance and advance with forearm crutches, a wheeled or platform walker.
Research articles:
- Utilizing Robotic Exoskeleton for Overground Walking in Acute and Chronic Stroke – Front. Neurorobot., 01 September 2021
Exoskeleton Report does not endorse one exoskeleton product over another. The exoskeleton catalog is purely for educational purposes. The catalog provides an easily accessible birds-eye view of the exoskeleton industry and a quick method to sort exoskeletons by type and purpose. All prices are approximate and are meant to provide a general sense of the cost of the devices.













Reviews
There are no reviews yet.