Description
Emovo Clinic is a one-handed robotic orthosis designed to support functional grasp during activities of daily living. It is intended for users with hand motor impairments following neurological accidents and can be used in clinical and domestic settings. The system assists active hand opening and closing, is adaptable to different hand morphologies, including varying finger lengths and diameters, and can be used on either the left or right hand.
The device uses a glove-less, soft-robotics approach with snap-button attachment and push-button control. Emovo Care describes the system as washable, spill resistant, and modular, with the ability to combine it with off-the-shelf splints such as thumb splints or wrist immobilizers. Movement force and speed can be personalized through an app, although the smartphone is not required during normal device operation.
It is built by Emovo Care, a startup based on technologies initially developed at the École Polytechnique Fédérale de Lausanne (EPFL) in Switzerland.
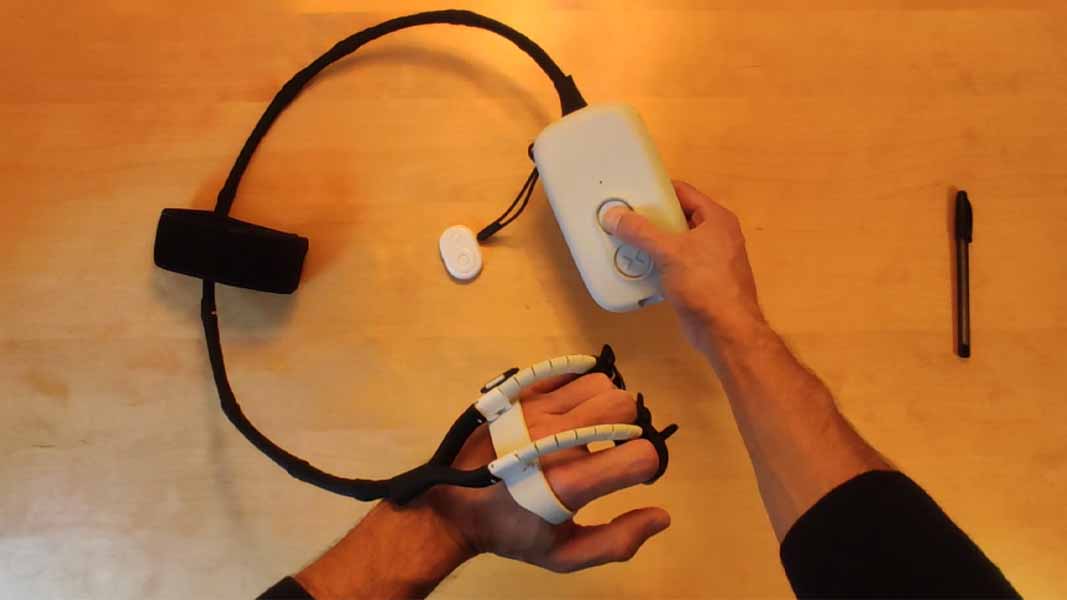
The exoskeleton consists of three parts:
– the actuation module: It includes the battery pack, motors, and input buttons. It can be handheld or worn at the waist.
– the tendons: 2 to control flexion and extension of 4 fingers (or 3 for the version with also thumb actuation).
– the hand interface: A partial glove that connects the tendons to the fingers through various anchoring points.
Application(s): Hand assistance; functional grasp; activities of daily living; neurorehabilitation / assistive use after neurological accidents
Exoskeleton Characteristics: Active hand opening and closing; push-button control; glove-less patented technology; snap-button attachment; adaptable to left or right hand; adaptable to different finger lengths and diameters; compatible with off-the-shelf splints; washable and spill resistant; force and speed customizable through an app; smartphone not required for normal operation.
Links to testimonials, independently produced videos by users or integrators, etc…:
Certifications and/or 3rd party testing results:
EN ISO 13485:2016, Certificate#: MD 736347
Emovo Care, see company profile
Exoskeleton Report does not endorse one exoskeleton product over another. The exoskeleton catalog is purely for educational purposes. The catalog is meant to provide an easily accessible birds-eye view of the exoskeleton industry, and a quick method to sort exoskeletons by type and purpose. All prices are approximate and are meant to provide a general sense of the cost of the devices.
This exoskeleton catalog entry has been made with the assistance of Stefano Carisi, MS in BioRobotics and Biomechanical Design from the Delft University of Technology an experienced product manager in human-machine collaboration.










Reviews
There are no reviews yet.