Description
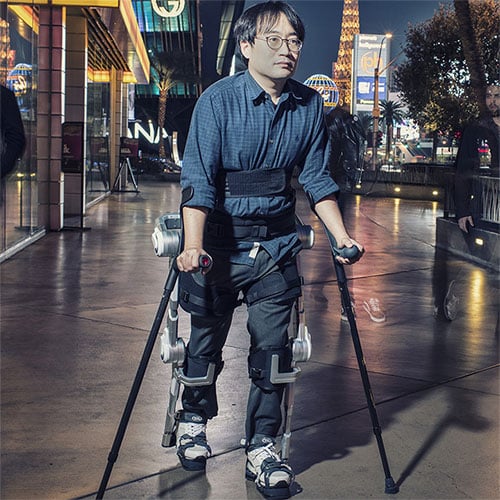
The H-MEX was demonstrated at the CES 2017 in Las Vegas. It joins the swelling ranks of Hyundai wearable robots. Both versions, H-MEX and HUMA are visibly more refined than previous medical exoskeletons by the car manufacturing giant.
According to the official company press release (link) Hyundai wants to expand its operations to include assistive mobility freedom devices.
Design specifications for HUMA:
- Metal frame that connects to the ground
- Powered hip-knee exoskeleton with as many as two motors per hip joint
- Support up to 40kg of wearer’s weight
- Running speed of 12km/h (very high speed for an assistive exoskeleton in 2017)
- Adjustable assistive torque based on an onboard motion control system
- Ability to sit, stand, move, turn, navigate stairs and run (note that as of the start of 2017, the HMEX and HUMA have not been approved by any regulatory bodies for these functions. It is very easy to confuse design intent and exoskeleton devices that have gone through FDA and similar testing and have been approved for such uses).
- Adjustable frame
- Rechargeable battery packs
- Artificial joint structure (a mixture of body adhering and end effector exoskeleton)
Check out these Exoskeleton Report articles involving Hyundai exoskeleton development.
Hyundai Motor Company, HQ: Seoul, South Korea, Link to full press release (and pictures) for the H-MEX, HUMA, and H-WEX https://www.hyundai.news/eu/articles/press-releases/hyundai-motor-leads-personal-mobility-revolution-with-advanced-robots.html
Exoskeleton Report does not endorse one exoskeleton product over another. The exoskeleton catalog is purely for educational purposes. The catalog is meant to provide an easily accessible birds-eye view of the exoskeleton industry, and a quick method to sort exoskeletons by type and purpose. All prices are approximate and are meant to provide a general sense of the cost of the devices.










Reviews
There are no reviews yet.