The Department of Veterans Affairs Palo Alto Health Care Systems (VAPAHCS) has submitted a request for an unidentified number of Ekso GT exoskeletons and a service contract for patients with stroke and spinal cord injury (SCI). Signed September 1st, 2016, the document titled Justification and Approval For Other Than Full and Open Competition (>$150K) includes the following:
- lists the capabilities of the Ekso GT
- explains why only the Ekso GT can be used to meet the needs of VAPAHCS
- provides an overview of the competition
- request to purchase several units (assuming one to three) and a four-year service contract
VAPAHCS has been researching rehabilitation exoskeletons and collecting information on the subject for some time now. VAPAHCS have already one Ekso GT on hand, and to our understanding in prior communication with the group, have hands-on experience with both the Indego by Parker Hannifin and ReWalk by ReWalk Robotics. Now that VAPAHCS is ready to make a purchase, we know how they are comparing Indego vs Ekso GT vs ReWalk exoskeletons.
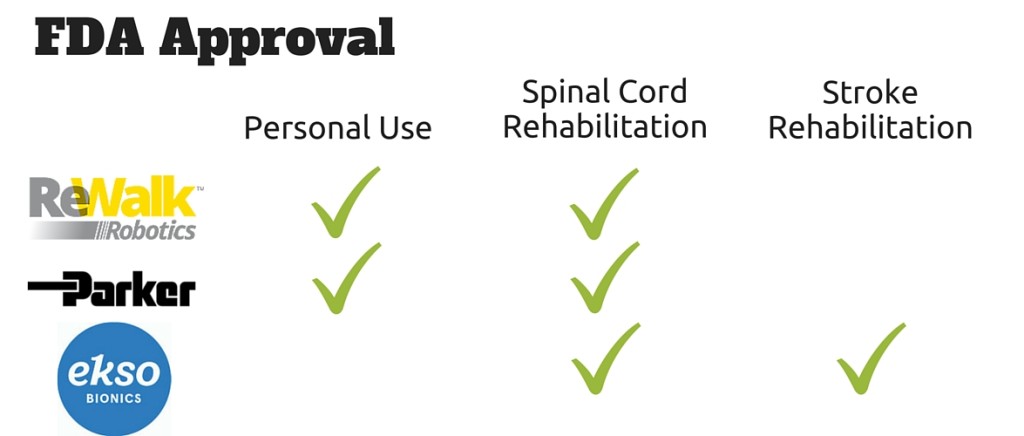
As of April 1st, 2016 there are three exoskeletons approved for use in the U.S. by the FDA. This creates a healthy competition between the exoskeleton companies. However, it has been difficult to determine how various government and private institutions view and differentiate the available wearable robots from one another (see: Commercial Exoskeletons From the Eyes of Physiotherapist, October 2015) The Justification and Approval document by VAPAHCS gives a glimpse into how that agency views the three FDA approved exoskeletons.
VAPAHCS considers the Ekso GT as uniquely qualified to fulfill its gait rehabilitation needs:
“Unique features of the Ekso GT Exoskeleton include:
- Provides the ability for the device to assist weak patients in walking with minimal arm and/or upper strength.
- Provides a variable and adaptive amount of power assistance on each leg ranging from 0-100%.
- Provides an integrated tether attachment that provides stability to patients with extreme mobility and/or balance impairment.
- Provides the ability for device software to use gait data gathered during each patient step to recalculate the assistance needed to apply to subsequent steps.
- Provides unlockable and adjustable abduction/adduction joints with an angle indicator.
- Provides unlockable thigh rotation joints.
- Compatible for use in a therapeutic environment by patients with mobility issues due to spinal cord injury up to C7 complete, any level of incomplete spinal cord injury, traumatic head injury, stroke, and many other debilitating causes.
- Provides four (4) different modes of use: trained spotter actuates steps with a button, user takes control of actuating steps with buttons on crutches or walker, user achieves the next step by moving their hips laterally and forward, and steps that are triggers by the users body shift and the initiation of forward leg movement.”
–VA Justification and Approval (J&A) For Other Than Full and Open Competition (>$150K)
This does not necessary mean that VAPAHCS interprets the Ekso GT as being a “superior” exoskeleton compared to its competitors, just uniquely suited for its gait rehabilitation needs. For example, if VAPAHCS was looking to purchase exoskeletons for home use, the Ekso GT would not even qualify for consideration because it is not presently approved for that purpose by the FDA.
According to the intent to purchase documentation, VAPAHCS views the ReWalk and Indego exoskeletons as “similar but not equivalent” when it came to gait rehabilitation. Here is how VAPAHCS view the two devices:
“Re Walk exoskeleton has FDA clearance, intended for patients with paraplegia due to spinal cord injuries at levels T7 (seventh thoracic vertebra) to L5 (fifth lumbar vertebra) when accompanied by a specially trained caregiver. It is also for people with spinal cord injuries at levels T4 (fourth thoracic vertebra) to T6 (sixth thoracic vertebra) where the device is limited to use in rehabilitation institutions. ReWalk is a motorized device worn over the legs and part of the upper body that helps an individual sit, stand, and walk with assistance from a trained companion, such as a spouse or home health aide. The battery-powered system features a light, wearable exoskeleton with motors at the hip and knee joints. The ReWalker controls movement using subtle changes in his/her center of gravity.
A forward tilt of the upper body is sensed by the system, which initiates the first step. Repeated body shifting generates a sequence of steps which mimics a functional natural gait of the legs.
Indego exoskeleton has FDA clearance, intended for patients paralyzed below the waist. Indego is strapped tightly around the torso, with rigid supports attaching to the hip, knee, and foot. Battery- powered, computer-controlled electric motors drive the joints, and the wearer navigates the device similar to a Segway, according to the engineers. Lean forward and the exoskeleton walks forward, lean back for a while and it will sit down. Indego has two features that are specifically designed to aid in rehabilitation:
The amount of robotic assistance adjusts automatically for users who have some muscle control in their legs. This allows them to use their own muscles while walking. When a user is totally paralyzed, the device does all the work. The other designs provide full power all of the time.
It is the only wearable robot that incorporates a proven rehabilitation technology called functional electrical stimulation. FES applies small electrical pulses to paralyzed muscles, causing them to contract and relax. FES can improve strength in the legs of people with incomplete paraplegia. For complete paraplegics, FES can improve circulation, change bone density and reduce muscle atrophy.”
Finally, VAPAHCS is not only looking to purchase Ekso GT unit(s), but also a service and support contract to go with them. Generally, robotic devices require a lot of care. Presenting the cost of ownership up front and providing a service package is an excellent move by Ekso Bionics. For more on the subject, see our article from July 2016 Will the exoskeleton companies turn a profit by selling a product or a service?
Sources:
65–Ekso GT Exoskeleton for Stroke & Spinal Cord Injury, FedBizOpps.Gov, September 1st, 2016
Special thanks to our contributor, Tim Retter for finding this information!











So in this kind of situation with VA could this help standardise technology within medical exoskeletons. I mean every area has an industry standard. Or are we just sitting on pure funtional use?
Excellent question! In this case, the VA is only a customer, and they are looking for what best fulfills their needs. However, there are government and non-government groups that are taking exoskeleton standardization (across all subfields, not just medical) very seriously.
According to your report, those 3(rewalk,indego,eksobionics)exoskeletons approved by the FDA. Do you have actual users’ reviews on those 3 exoskeletons? Plus, which one do you think should be the best? or which one users are satisfied with the most? Please let me know as specific as you can.
I think like the FDA report states, it is not possible to directly compare one exoskeleton to another in a head-to-head competition. For a specific application, one might be better than the other. And the applications can vary: left-right paralysis, lower body paralysis at various spine levels, incomplete/complete, the user already had physical therapy…
There are people that have used all three devices extensively, and they have never revealed a favorite of one over the other. The best approach would be to evaluate each depending on your specific need and application. Here is an easy example of what I mean. Say you are a medical care provider and you want to use the exoskeleton on five or more patients during the day. The ease with each the exo can be adjusted to each user is important. But if you were looking for a personal device for one user, then the speed of fit adjustment becomes irrelevant.
Are these open to the public to buy or are they only available to the VA?
If so, how much does an exoskeleton cost? Would it be covered by health insurance?
Yes, these are open to the public to purchase, they are not covered by insurance companies, and they cost upward of $50,000.
Are this devices a possible tool also for people with SMA type 3 (able to stand up and walk for few minutes?)
Thank you!
I would venture to say that the ReWalk is, but I would connect with the manufacturers first if I were you.