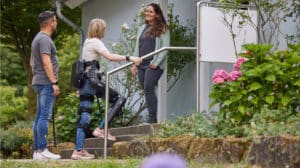Developing news: ReWalk Robotics, now operating under the name Lifeward, and Ekso Bionics, have both separately announced that the Centers for Medicare &...
Tag - ReWalk Robotics
ReWalk Robotics has overcome a major regulatory hurdle by having the first medical exoskeleton to have FDA approval for assisting with walking up stairs and...
ReWalk Robotics, one of the leaders in medical powered exoskeletons and exosuits, has released its three-month and six-month updates for 2021. The company...
In addition to ErgoX 2020 and WeRob/WearRAcon Europe 20, a myriad of exoskeleton companies and organizations are offering a plethora of webinars this summer...
In an industry first, the ReWalk Personal exoskeleton has been granted a CMS HCPCS Level II code. In addition to being a success for ReWalk Robotics, this is a...
ReWalk Robotics has been at the forefront of negotiating insurance coverage for medical exoskeletons, especially its ReWalk 6.0 powered walking assist device...
The ReWalk Robotics stroke rehabilitation exosuit, ReStore Exo-Suit, has been cleared by the U.S. Food and Drug Administration (FDA) for sale to rehabilitation...
ReWalk Robotics (Nasdaq: RWLK) best known for developing and offering the ReWalk walking assist exoskeleton is on track to become even more famous with a new...
The wearable robotics giant ReWalk Robotics (Nasdaq: RWLK) executed a one to twenty-five reverse share split on April 1st of 2019, and no, it is not an April...
ReWalk Robotics has issued a press release that Cigna, a global health service company, has already revised its policy on medical exoskeleton coverage for...